
Protein Thioester Synthesis Enabled By: Sortase
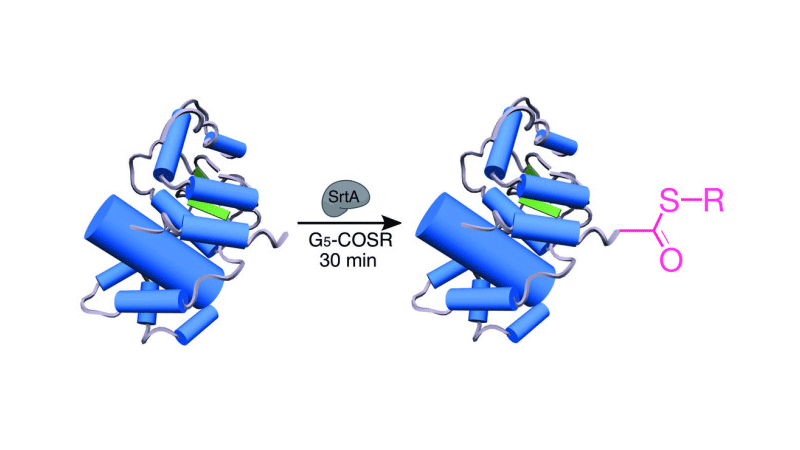
Protein Thioester Synthesis Enabled By: Sortase
J. Am. Chem. Soc., 2012, 134:10749-52
Published
Published online Jun 19, 2012
Abstract
Proteins containing a C-terminal thioester are important intermediates in semisynthesis. Currently there is one main method for the synthesis of protein thioesters that relies upon the use of engineered inteins. Here we report a simple strategy, utilizing sortase A, for routine preparation of recombinant proteins containing a C-terminal αthioester. We used our method to prepare two different anthrax toxin cargo proteins: one containing an αthioester and another containing a D-polypeptide segment situated between two protein domains. We show that both variants can translocate through protective antigen pore. This new method to synthesize a protein thioester allows for interfacing of sortase-mediated ligation and native chemical ligation.



