Monitoring the Kinetics of the pH-Driven Transition of the Anthrax Toxin Prepore to the Pore by Biolayer Interferometry and Surface Plasmon Resonance.
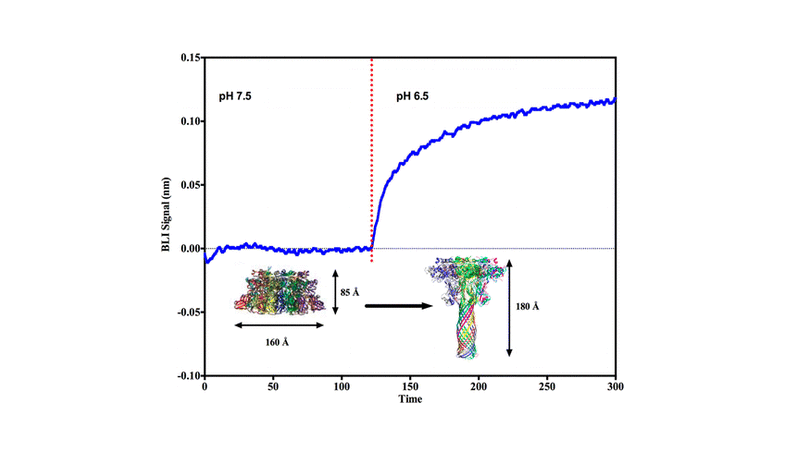
Domain 2 of the anthrax protective antigen (PA) prepore heptamer unfolds and refolds during endosome acidification to generate an extended 100 Å β barrel pore that inserts into the endosomal membrane. The PA pore facilitates the pH-dependent unfolding and translocation of bound toxin enzymic components, lethal factor (LF) and/or edema factor, from the endosome to the cytoplasm. We constructed immobilized complexes of the prepore with the PA-binding domain of LF (LFN) to monitor the real-time prepore to pore kinetic transition using surface plasmon resonance and biolayer interferometry (BLI). The kinetics of this transition increased as the solution pH was decreased from 7.5 to 5.0, mirroring acidification of the endosome. Once it had undergone the transition, the LFN-PA pore complex was removed from the BLI biosensor tip and deposited onto electron microscopy grids, where PA pore formation was confirmed by negative stain electron microscopy. When the soluble receptor domain (ANTRX2/CMG2) binds the immobilized PA prepore, the transition to the pore state was observed only after the pH was lowered to early (pH 5.5) or late (pH 5.0) endosomal pH conditions. Once the pore formed, the soluble receptor readily dissociated from the PA pore. Separate binding experiments with immobilized PA pores and the soluble receptor indicate that the receptor has a weakened propensity to bind to the transitioned pore. This immobilized anthrax toxin platform can be used to identify or validate potential antimicrobial lead compounds capable of regulating and/or inhibiting anthrax toxin complex formation or pore transitions.
Pentelute Lab, MIT, Publications, Brad Pentelute, Science, Bio, Chemistry, Peptide, Research,
15743
portfolio_page-template-default,single,single-portfolio_page,postid-15743,bridge-core-3.0.1,qode-page-transition-enabled,ajax_fade,page_not_loaded,,paspartu_enabled,paspartu_on_top_fixed,paspartu_on_bottom_fixed,qode_grid_1200,qode_popup_menu_push_text_top,qode-theme-ver-28.7,qode-theme-bridge,disabled_footer_top,wpb-js-composer js-comp-ver-6.8.0,vc_responsive
Monitoring the Kinetics of the pH-Driven Transition of the Anthrax Toxin Prepore to the Pore by Biolayer Interferometry and Surface Plasmon Resonance.
Monitoring the Kinetics of the pH-Driven Transition of the Anthrax Toxin Prepore to the Pore by Biolayer Interferometry and Surface Plasmon Resonance.
Naik S, Brock S, Akkaladevi N, Tally J, McGinn-Straub W, Zhang N, Gao P, Gogol EP, Pentelute BL, Collier RJ, Fisher MT., Biochemistry. 2013 Sep 17;52(37):6335-47
Published
Online September, 17 2013
Abstract
Domain 2 of the anthrax protective antigen (PA) prepore heptamer unfolds and refolds during endosome acidification to generate an extended 100 Å β barrel pore that inserts into the endosomal membrane. The PA pore facilitates the pH-dependent unfolding and translocation of bound toxin enzymic components, lethal factor (LF) and/or edema factor, from the endosome to the cytoplasm. We constructed immobilized complexes of the prepore with the PA-binding domain of LF (LFN) to monitor the real-time prepore to pore kinetic transition using surface plasmon resonance and biolayer interferometry (BLI). The kinetics of this transition increased as the solution pH was decreased from 7.5 to 5.0, mirroring acidification of the endosome. Once it had undergone the transition, the LFN-PA pore complex was removed from the BLI biosensor tip and deposited onto electron microscopy grids, where PA pore formation was confirmed by negative stain electron microscopy. When the soluble receptor domain (ANTRX2/CMG2) binds the immobilized PA prepore, the transition to the pore state was observed only after the pH was lowered to early (pH 5.5) or late (pH 5.0) endosomal pH conditions. Once the pore formed, the soluble receptor readily dissociated from the PA pore. Separate binding experiments with immobilized PA pores and the soluble receptor indicate that the receptor has a weakened propensity to bind to the transitioned pore. This immobilized anthrax toxin platform can be used to identify or validate potential antimicrobial lead compounds capable of regulating and/or inhibiting anthrax toxin complex formation or pore transitions.
Category
2013, Publications