
Convergent Diversity-Oriented Side-Chain Macrocyclization Scan for Unprotected Polypeptides
Convergent Diversity-Oriented Side-Chain Macrocyclization Scan for Unprotected Polypeptides
Zou, Spokoyny, Zhang, Simon, Yu, Lin, Pentelute, Org. & Biomol. Chem., 2014, 12, 566-573.
Published
Online January 28, 2014
Abstract
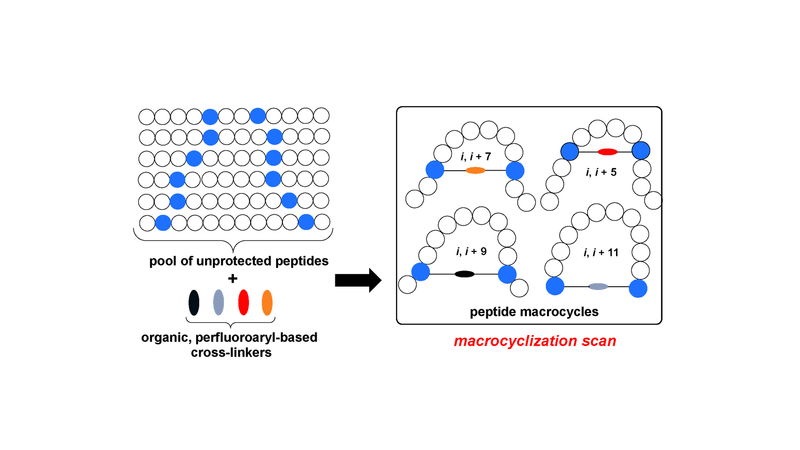
Here we describe a general synthetic platform for side-chain macrocyclization of an unprotected peptide library based on the SNAr reaction between cysteine thiolates and a new generation of highly reactive perfluoroaromatic small molecule linkers. This strategy enabled us to simultaneously "scan" two cysteine residues positioned from i, i + 1 to i, i + 14 sites in a polypeptide, producing 98 macrocyclic products from reactions of 14 peptides with 7 linkers. A complementary reverse strategy was developed; cysteine residues within the polypeptide were first modified with non-bridging perfluoroaryl moieties and then commercially available dithiol linkers were used for macrocyclization. The highly convergent, site-independent, and modular nature of these two strategies coupled with the unique chemoselectivity of a SNAr transformation allows for the rapid diversity-oriented synthesis of hybrid macrocyclic peptide libraries with varied chemical and structural complexities.



