
Conformational Stabilization and Rapid Labeling of a 29-Residue Peptide by a Small Molecule Reaction Partner
Conformational Stabilization and Rapid Labeling of a 29-Residue Peptide by a Small Molecule Reaction Partner
Biochemistry201958101343-1353
Publication Date:February 6, 2019
https://doi.org/10.1021/acs.biochem.8b00940
Ethan D. EvansZachary P. GatesZhen-Yu J. SunAlexander J. Mijalis, Bradley L. Pentelute*
Abstract
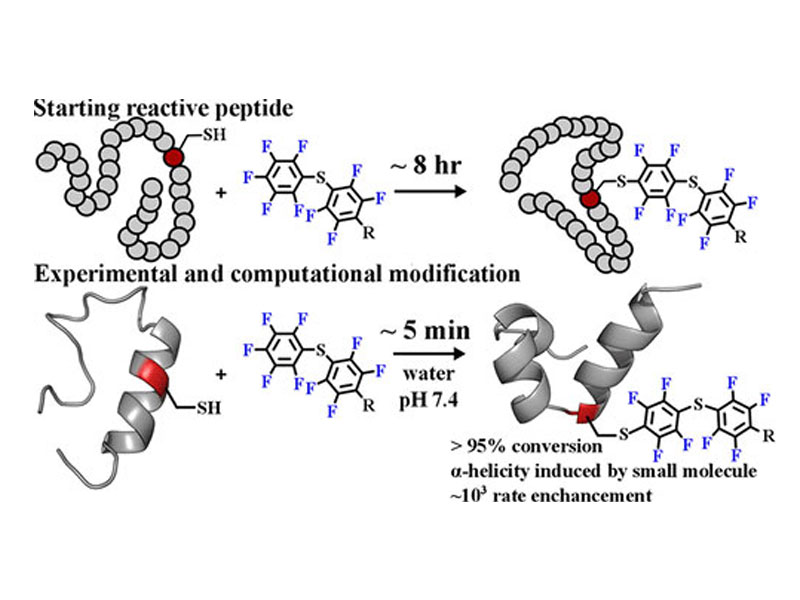
A 29-residue peptide (MP01), identified by in vitro selection for reactivity with a small molecule perfluoroaromatic, was modified and characterized using experimental and computational techniques, with the goal of understanding the molecular basis of its reactivity. These studies identified a six-amino acid point mutant (MP01-Gen4) that exhibited a reaction rate constant of 25.8 ± 1.8 M–1 s–1 at pH 7.4 and room temperature, approximately 2 orders of magnitude greater than that of its progenitor sequence and 3 orders of magnitude greater than background cysteine reactivity. MP01-Gen4 appeared to be conformationally dynamic and exhibited several properties reminiscent of larger protein molecules, including denaturant-sensitive structure and reactivity. We believe the majority of the reaction rate enhancement can be attributed to interaction of MP01-Gen4 with the perfluoroaromatic probe, which was found to stabilize a helical conformation of both MP01-Gen4 and nonreactive Cys-to-Ser or Cys-to-Ala variants. These findings demonstrate the ability of dynamic peptides to access proteinlike reaction mechanisms and the potential of perfluoroaromatic functionality to stabilize small peptide folds.



