
Arylation Chemistry for Bioconjugation
Arylation Chemistry for Bioconjugation
First published: 06 November 2018 https://doi.org/10.1002/anie.201806009
Dr. Chi Zhang Dr. Ekaterina V. Vinogradova Prof. Dr. Alexander M. Spokoyny Prof. Dr. Stephen L. Buchwald Prof. Dr. Bradley L. Pentelute
Abstract
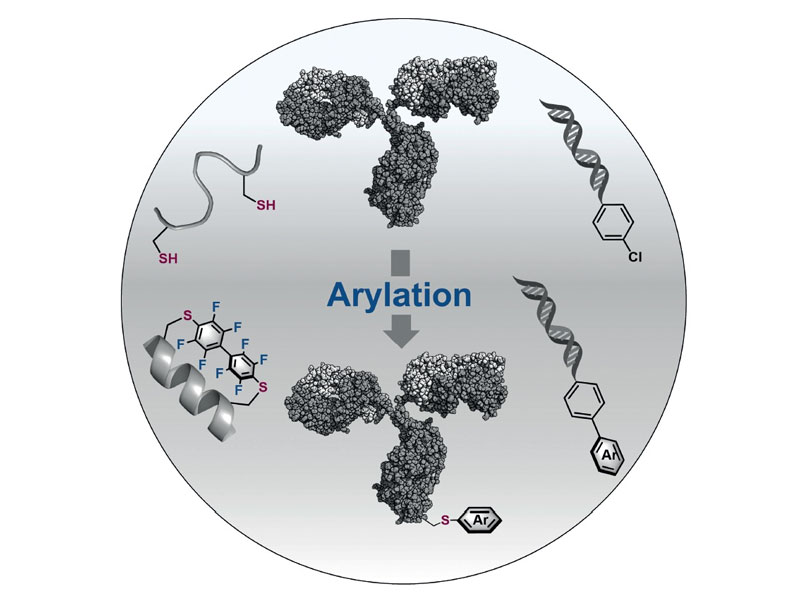
Bioconjugation chemistry has been used to prepare modified biomolecules with functions beyond what nature intended. Central to these techniques is the development of highly efficient and selective bioconjugation reactions that operate under mild, biomolecule compatible conditions. Methods that form a nucleophile–sp2 carbon bond show promise for creating bioconjugates with new modifications, sometimes resulting in molecules with unparalleled functions. Here we outline and review sulfur, nitrogen, selenium, oxygen, and carbon arylative bioconjugation strategies and their applications to modify peptides, proteins, sugars, and nucleic acids



